Fuel cells are a remarkable source of clean energy that operate at high electrical efficiency and release very few emissions or pollutants into the atmosphere. The low emissions and high efficiency of fuel cells are key to understanding the fuel cell value proposition and sustainability profile.
Are fuel cells efficient?
FuelCell Energy’s carbonate fuel cell power plants have superior fuel efficiency to comparably sized combustion-based power systems, because of the more direct conversion of fuel energy to electrical energy. Less fuel is needed to produce power in a high efficiency fuel cell, resulting in a lower total operating cost. Low fuel consumption also results in reduced CO2 emissions if the fuel is nonrenewable, such as natural gas.
Fuel cells efficiently extract power from fuel through an electrochemical reaction and avoid the energy losses and emissions associated with combustion and mechanical conversion to electricity. High temperature fuel cells (such as carbonate fuel cells) can reach more than 60 percent efficiency and outperform similar-sized conventional energy sources like combustion engines and gas turbines.
Fuel cell efficiency vs. combustion
For a combustion process to generate electricity from a fuel, it must first convert the fuel's chemical energy into heat, then convert the heat to mechanical work, and lastly convert the mechanical work into electricity. Each additional step causes the overall efficiency to drop. A fuel cell (or any electrochemical system) directly converts the chemical energy stored in fuels to electricity, skipping the intermediate steps that result in energy losses.
High-temperature fuel cells like those used in FuelCell Energy’s platforms typically operate at higher efficiencies than low-temperature fuel cells. This is due to their ability to use their internal heat generation for hydrogen production through steam reforming of hydrocarbon fuels. Carbonate fuel cells have the additional advantage of producing high-quality heat that can be harnessed to create useable resources like high-pressure steam.
High-temperature fuel cells are also capable of processing a broader range of fuels, including biogas, natural gas, and propane. These fuels are processed into hydrogen through the steam-reformation process – an endothermic reaction sustained by the heat generated by the fuel cell. One cubic foot of natural gas creates an equivalent of four cubic feet of hydrogen, which will carry a greater heating value than the original gas.
In cogeneration systems, fuel cells can produce power and useful outputs like thermal energy or cooling from a single source of energy. If waste heat is captured through a combined heat and power system (CHP), the rate of efficiency is much higher. FuelCell Energy’s high temperature power plants are uniquely suited to provide high-quality, useful thermal energy which avoids the use of fuel in on-site boilers or chillers. This further reduces emissions on top of the emissions reductions stemming from the high electrical efficiency.
Fuel cell combined heat and power systems (CHP)
Fuel cells operate at high enough temperatures to efficiently complete the chemical reaction taking place inside the stack. For example, a molten carbonate fuel cell might operate at a temperature of more than one thousand degrees Fahrenheit. To maximize the fuel cell’s efficiency, combined heat and power (CHP) systems use extra energy by recovering heat that might otherwise be lost into the atmosphere.
Fuel cell combined heat and power systems use a heat exchanger, or heat recovery unit, to recover energy from the exhaust downstream of the fuel cell, transitioning that heat to useful energy. Using the exhaust heat generated by the typical operation of the system increases the net energy output of the plant and helps achieve a higher plant efficiency.
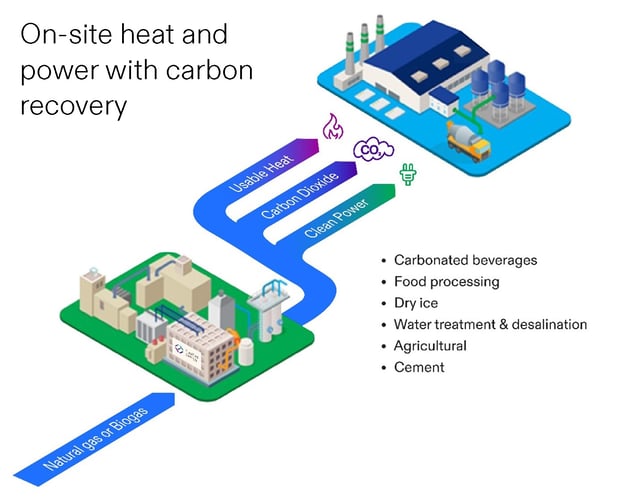
The heat generated by a fuel cell CHP system can be used in a variety of ways. Food producers can use heat from the fuel cell to support bakery operations, reducing the fuel needs for the plant’s boilers. Universities have used heat for their buildings and pools. Wastewater treatment facilities use heat from the fuel cells to support anaerobic digestion. Industrial facilities have used fuel cell waste heat for steam production. In all cases the fuel cell waste heat avoids the use of a combustion fuel in a boiler or other process, avoiding carbon and criteria emissions.
What is LHV and HHV?
Lower heating value (LHV) and higher heating value (HHV) are values that define the energy content of a fuel. These values are typically used to describe a fuel cell’s electrical operating efficiency, or the system’s overall thermal efficiency.
Lower Heating Value refers to the amount of energy theoretically released if you burn the fuel and produce water vapor. There is theoretical energy that can be obtained by condensing the water vapor to liquid - when this theoretical energy is added to the LHV, the result is the Higher Heating Value.
For example, the theoretical LHV of methane is 910 British thermal unit (Btu) per standard cubic foot (or 0.267 kWh per cubic foot). The HHV of methane is 1009 Btu per standard cubic foot (or 0.296 kWh per cubic foot). They are two different ways of describing the energy content of methane.
Gas suppliers typically use HHV when describing the amount of fuel provided or the price of fuel, which is typically described in dollars per million Btu, and the Btu is based on HHV. Power generators typically use LHV to describe the efficiency of their systems. Gas suppliers use HHV to imply that more energy is being provided while power generation equipment suppliers use LHV to imply that less energy is needed to produce power. LHV and HHV are two ways of describing the same thing (the energy content of a fuel), but it is important to specify which is being used when describing efficiency or fuel use.
How to calculate fuel cell efficiency (LHV or HHV)
Electrical efficiency is calculated by taking the measured net AC electrical output from the fuel cell power plant and deducting any internal parasitic loads that reduce the exported power. This value represents the net AC electrical power produced by the fuel cell plant.
Fuel cell plant efficiency is determined by dividing the amount of net AC energy provided by the plant over a period (in kWh, calculated by multiplying average kW output by the hours in the period) by the total fuel supplied over the period, in kWh (the average kW fuel flow times the hours in the period). If the fuel kW used in the calculation is the 0.267 kWh per cubic foot LHV, then the LHV efficiency has been calculated. If the fuel kW used in the calculation is the 0.296 kWh per cubic foot HHV, then the HHV efficiency has been calculated.
Read more about how a fuel cell works or contact us today to speak with our team.

.png?width=1037&height=559&name=fce-data-center-whitepaper-graphic%20(1).png)